 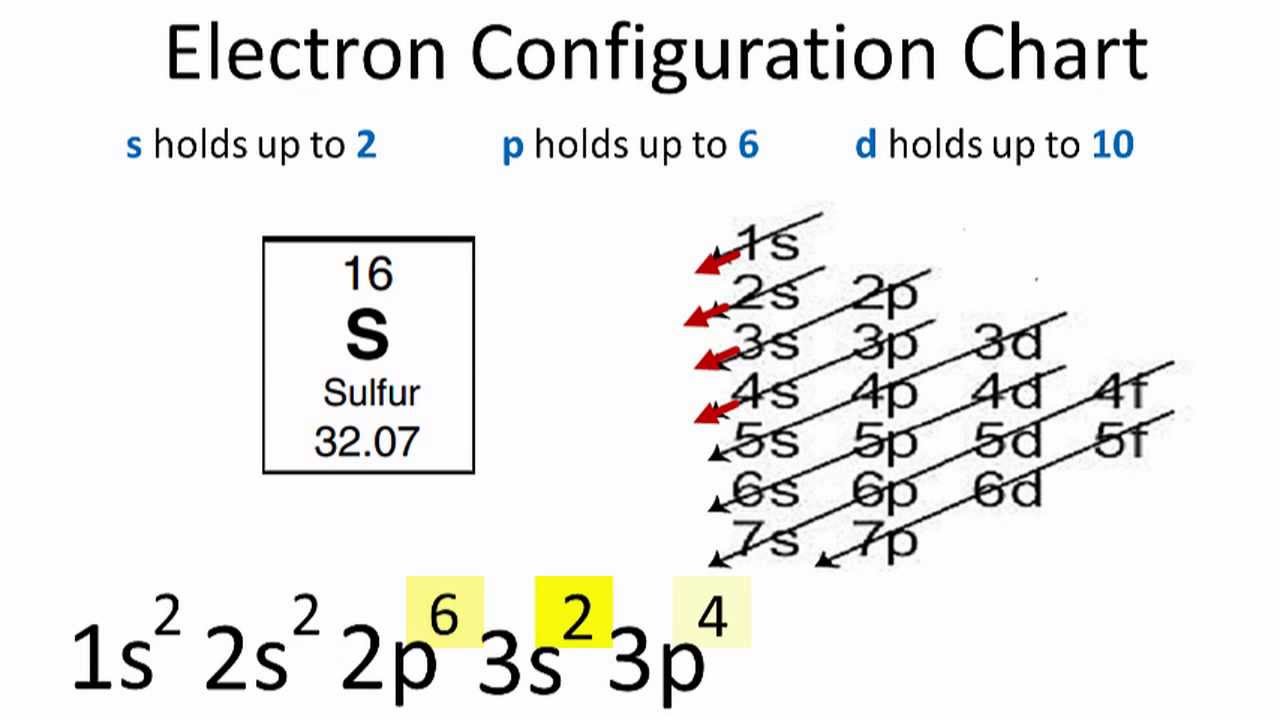 
It discusses the molecular geometry, bond angle, hybridization. This chemistry video tutorial explains how to draw the lewis structure of SO3 also known as Sulfur Trioxide. What is the Difference Between SO2 and SO3 – Comparison of Key Differences58K views 2 years ago. What is SO3 – Definition, Chemical Structure and Properties, Sulfuric Acid Production 3. What is SO2 – Definition, Chemical Structure and Properties, Oxidation State 2. SO3 is also a nonpolar molecule due to its trigonal planar. Is SO3 a polar or non polar? There are three molecules with S-O bonds and no lone pair of electrons in the central sulfur atom, sulfur trioxide (SO3) is a nonpolar molecule. The shape of most molecules can be predicted using the Valence Shell Electron Pair Repulsion (VSEPR) method.The molecular geometry of SO3 is trigonal planar with symmetric charge distribution on the central atom. It is determined by the central atom and the surrounding atoms and electron pairs. Molecular geometry is the 3-dimensional shape that a molecule occupies in space. This arrangement, or 3-dimensional geometric layout, of the bonded atoms (and electrons) in a molecule is termed as its molecular geometry.Molecular Geometry. Draw the Lewis structure and write the molecular geometry and hybridization on the central atom, and …Molecular Geometry: The different atoms in a molecule have a geometric arrangement in space around the central atom. For this molecule, determine the molecular geometry, electron domain geometry, bond angles, and hybridization about the central atom. ClO3- appears to be a wet substance mostly a solid which has. 
The molecular weight of the compound is 83.45 g/mol. Chlorine trioxide is non-combustible in nature but when it gets combined with combustible materials explosion is likely to occur. ClO3- or chlorine trioxide or chlorate is a monovalent inorganic anion that is soluble in water. The first step is to sketch the molecular geometry of the SO2 molecule, to calculate the lone pairs of the electrons in the central sulfur and terminal oxygen atoms the second step is to calculate the SO2 hybridization, and the third step is to give perfect notation for the SO2. A three-step approach for drawing the SO2 molecular can be used. Note that the precise angle is 106.0 degrees).The dot structure for sulfur dioxide has sulfur with a double bond to an oxygen on the left, and two lone pairs of electrons on that oxygen, and the sulfur with a double bond to an oxygen on the right, and two lone pairs of electrons on that oxygen. A quick explanation of the molecular geometry of SO32- including a description of the SO32- bond angles. Draw the Lewis structure for CCl2F2 (carbon is the central atom) and determine the following: a. Predict the electron geometry and molecular geometry and state whether the molecule is polar or nonpolar. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
